Research Overview
The Dudley laboratory is focused on:
- Mechanisms of virus-induced diseases, including breast cancer and lymphomas
- Retrovirus replication and development of efficient viral vectors
- Viral manipulation of cellular processes, such as innate immunity mediated by Apobec cytidine deaminases and ER-associated degradation (ERAD)
Unlike human complex viruses, such as HIV-1 or human T-cell leukemia virus type 1 (HTLV-I), mouse mammary tumor virus (MMTV) provides a model system that allows biochemical, genetic, and biological studies of viral pathogenesis. Our early studies revealed that the types of tumors induced by MMTV depended on cell-type specific enhancers within the viral transcriptional regulatory region (LTR). Substitution of the MMTV enhancer region switched virus-induced disease from breast cancers to T-cell lymphomas. We also showed that MMTV encodes several different regulatory and accessory proteins that contribute to viral replication and tumorigenesis, including superantigen (Sag), Rem, signal peptide (SP), and Rem-CT. Accessory proteins only are required during MMTV replication in mice.
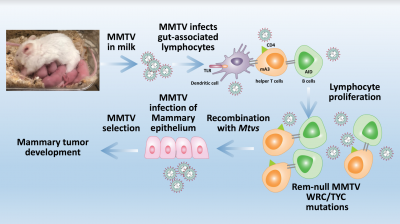
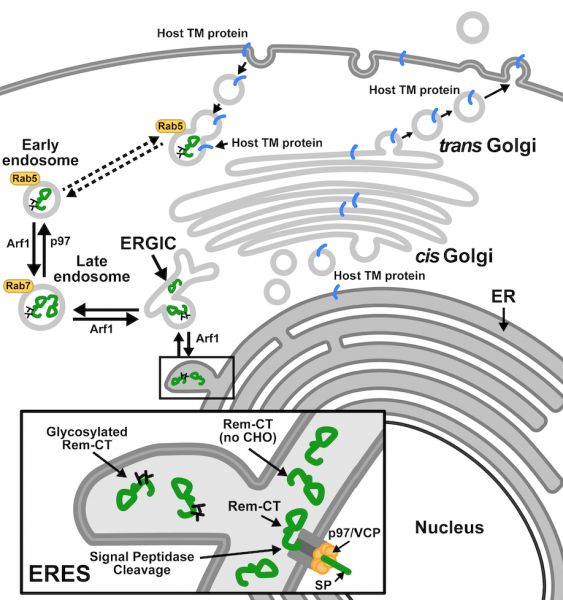
We identified the virally encoded protein, Rem (regulator of MMTV mRNA export and expression). Rem is a fascinating protein with a highly unusual trafficking scheme and multiple functions. Rem is translated as a precursor at the ER membrane and cleaved into a functional signal peptide (SP) and Rem-CT. SP is actively retrotranslocated to the cytosol and then imported into the nucleus for MMTV RNA export and translation. Our work showed that SP retrotranslocation is dependent on the essential AAA ATPase VCP/p97, but avoids ubiquitylation and ERAD for nuclear function [see animation: Rem_processing.mov]. We also demonstrated that Rem-CT (Rem C-terminal cleavage product) traffics from the ER to the cell surface through endosomes using VCP/p97 without passing through the Golgi network. The function of this cellular pathway and Rem-CT protein have yet to be determined. Rem precursor activity was identified by analysis of loss-of-function mutants in mice. Rem-null mutants showed increased cytidine mutations within viral DNA, which were typical of the Apobec family member, activation-induced cytidine deaminase (AID), but AID-knockout mice did not. Our data suggest that Rem precursor is the first known protein inhibitor of AID, which functions in antiviral innate immunity.
In addition, our laboratory is interested in improving retroviral vectors for molecular biology and gene therapy. Recent work indicates that unintegrated retroviral DNA activates a cellular signaling pathway that results in increased cap-dependent viral translation. Together, our experiments reveal how retroviruses provide the means for understanding basic cell biology with applications to disease treatments.


