LCID's Jason Upton Discovers How Cells Recognize Influenza Virus
Influenza virus infects millions of people and causes nearly half a million deaths per year worldwide (WHO Ref. 1). As flu season approaches, UT-Austin Researcher Jason Upton, with collaborators at Fox Chase Cancer Center, St. Jude’s Children’s Hospital and Icahn School of Medicine, has uncovered an unexpected piece in the puzzle of how host organisms detect and respond to influenza.
Influenza A virus, a virus that uses RNA to pass along its genetic information and propagate itself, replicates in epithelial cells of the airway in the respiratory track. The replicating virus can damage airways and compromise lung function, resulting in the symptoms of respiratory illness and inflammation associated with flu. If the host immune system isn’t able to control the virus, this can be fatal.
Dr. Upton and colleagues are looking closely at what happens on a molecular level when Influenza A infects a mammalian cell. Previous work had shown that Influenza A virus infection activates a key protein kinase called RIPK3, triggering two crucial cell death pathways, (1) necroptosis and (2) apoptosis (Nogusa et al., 2016). These pathways lead to the efficient ‘suicide’ of infected cells, which prevents them from becoming virus factories that overwhelm and kill the host. However, crucial details on how RIPK3 was activated remained a mystery.
In their most recent publication, these researchers have identified the cellular host protein that allows cells to detect the presence of influenza virus and self-destruct (Thapa et al., 2016). The protein is called DNA-dependent Activator of Interferon regulator factors, or DAI for short. As its name suggests, DAI was initially described as a sensor for DNA that escaped its normal compartment, the nucleus. It has also been shown to activate RIPK3-dependent cell death in response to some viruses with DNA genomes, such as herpesviruses (Upton et al., 2012). However, after ruling out more likely candidates, the team tested whether DAI also plays an important role linking influenza, an RNA virus, to the activation of RIPK3.
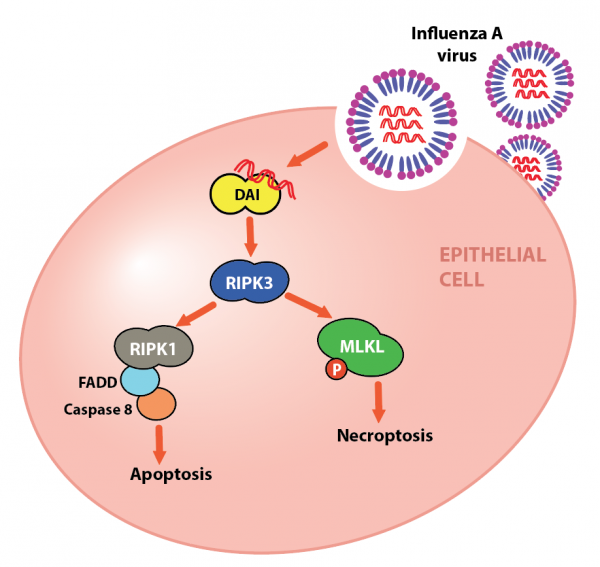
The group showed that, during infection, DAI recognizes and binds directly to Influenza A RNA genomes and this recognition allows it to associate with, and activate RIPK3. In turn, the kinase induces necroptosis and apoptosis through the effector proteins MLKL or RIPK1, respectively. The team demonstrated this in cultured cells, showing that cells lacking DAI are resistant to both cell death pathways triggered by Influenza A, as well as showing that mice lacking DAI are unable to control Influenza A replication and succumb to lethal respiratory infection by this virus. Therefore, DAI’s role as an RNA sensor in mammalian cells is important for survival of flu virus infection. It is the critical missing puzzle piece between Influenza A replication and RIPK3 activation, and it provides exciting new avenues for further research and potential therapeutic strategies.
References:
1. World Health Organization (WHO), “Influenza (seasonal)” (Fact sheet no. 211, WHO, 2014)
Further reading:
1. DAI / ZBP1