Research
Electrostatic fields at protein-protein interfaces
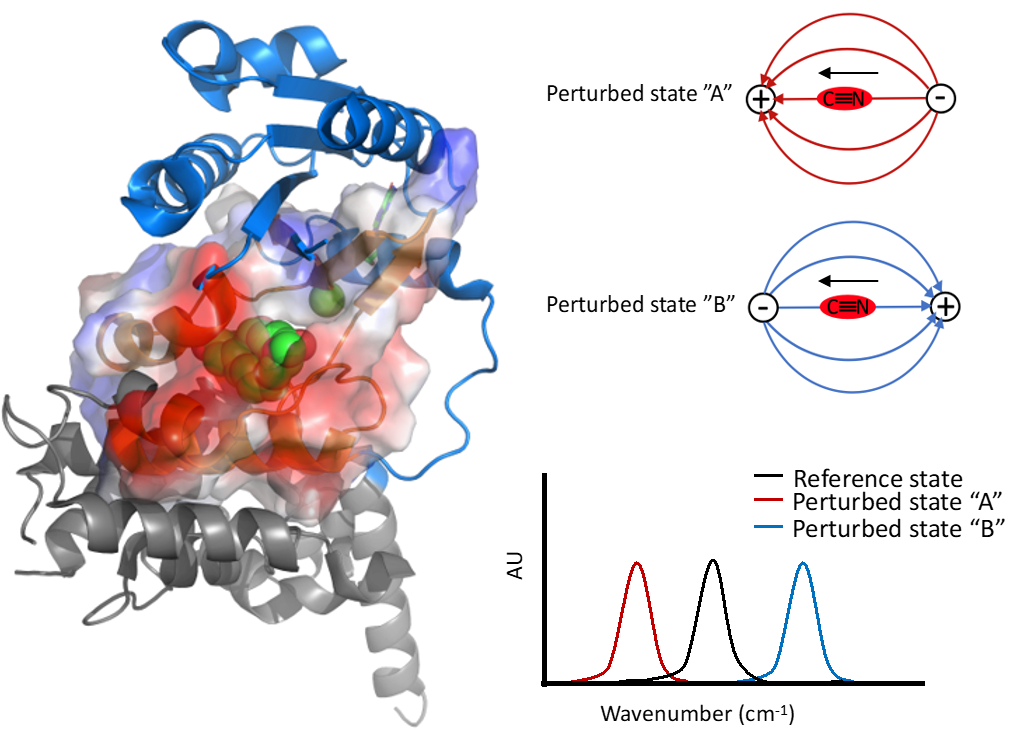
The affinity and specificity of macromolecular interactions are the result of both structural and electrostatic driving forces, but while the field of structural biology has made great advances, much less is understood about electrostatic influences. We measure the electrostatic fields at protein-protien intrafaces using vibrational Stark effect (VSE) spectroscopy and seek to develop computational models that accurately predict these interactions.
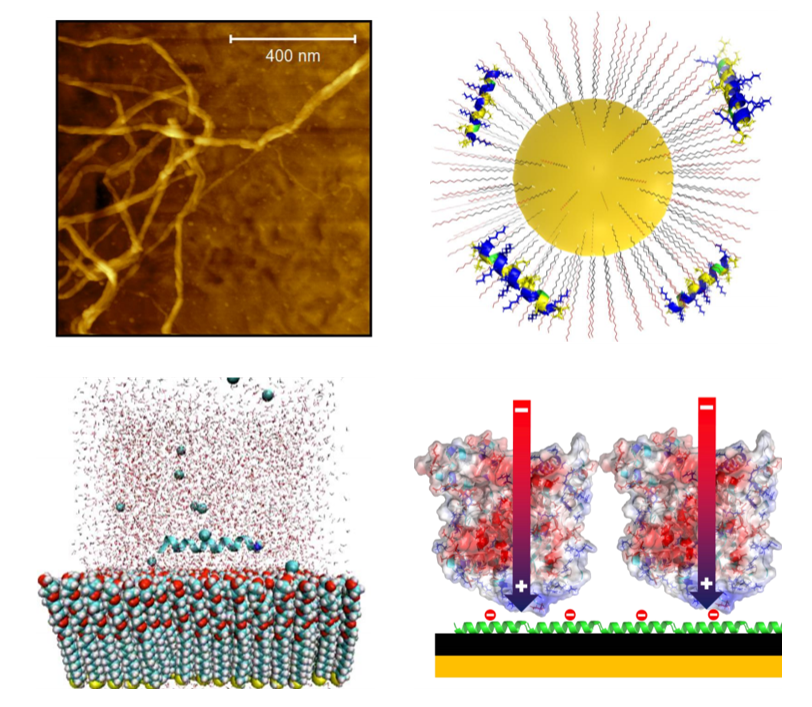
Proteins provide an extraordinary range of structures, properties, and functions that could be exploited for novel devices and materials if the biological molecule could be effectively integrated with standard inorganic platforms and devices. We are developing surface chemical functionalization techniques that use noncovalent binding of proteins through long-range electrostatic forces that mimic biological protein-protein interaction mechanisms.
Molecular Dynamics Simulations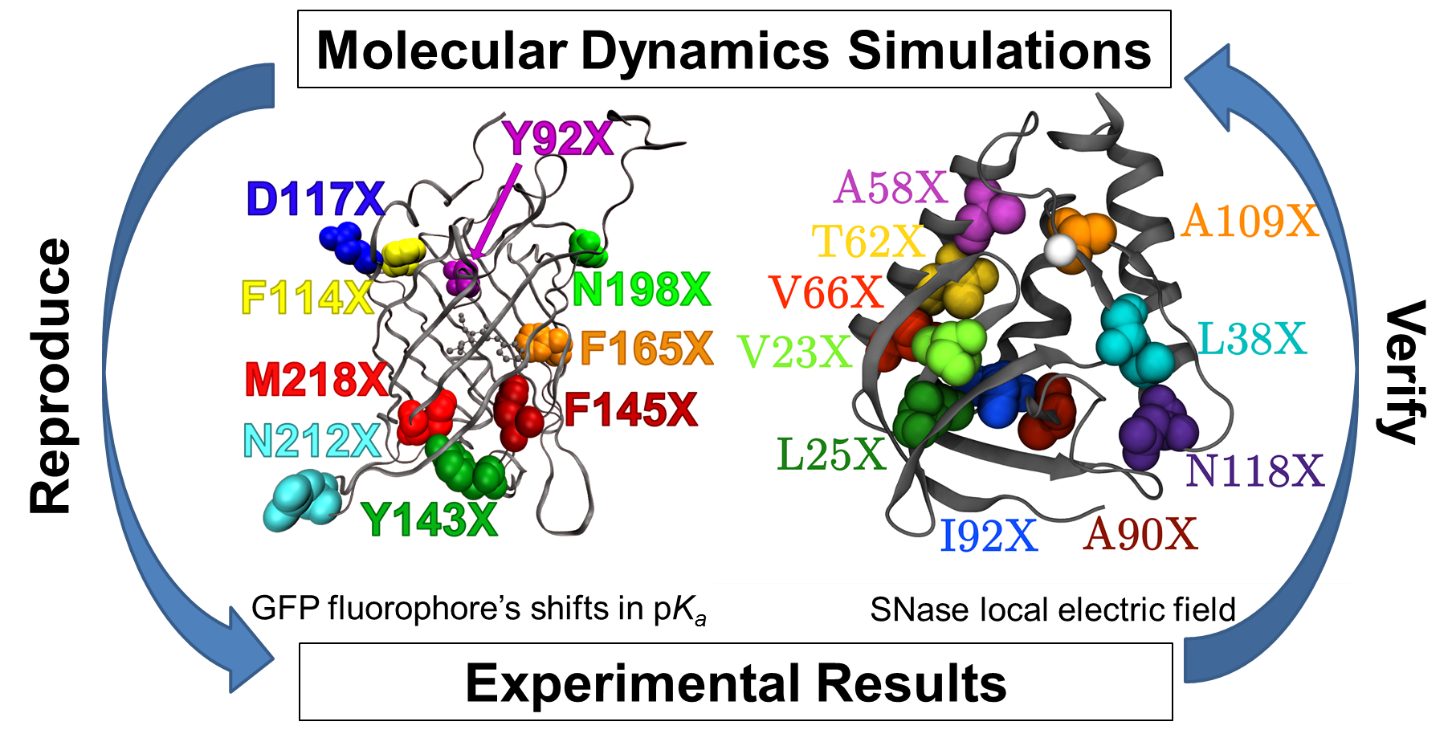
All experimental systems investigated in our laboratory are explored through computational modeling and simulation. We use molecular dynamics (MD) simulations to model proteins and peptides in order to understand electrostatic interactions on a molecular level. We work with multiple levels of theory to testing and develop new electrostatic modeling methodologies. We then use computational modeling to predict and guide new experiments.
Understanding fundamental photophysics of the GFP chromophore through experiment and theory
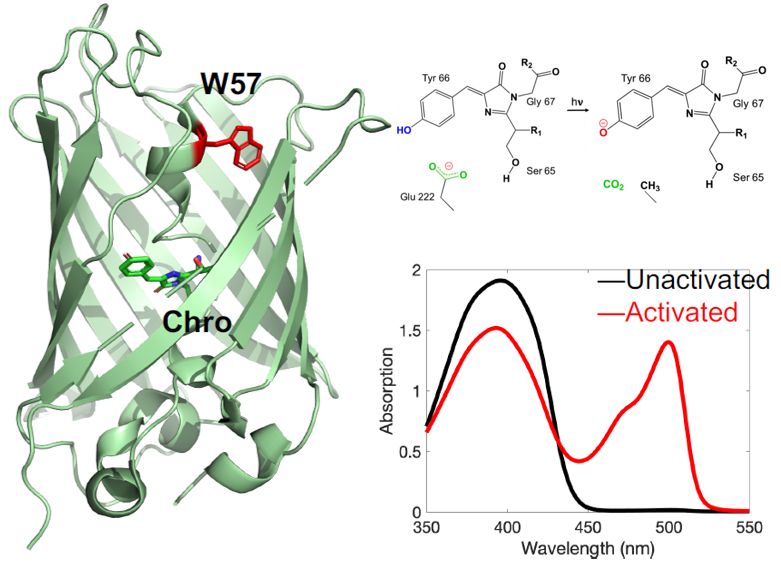
It is impossible to overstate the importance of GFP in modern biophysical and biomedical research. Although a number of GFP constructs for imaging biological details within live cells have been demonstrated, there are significant gaps in our fundamental understanding of useful chromophore properties that impede more sophisticated applications of GFP in biological research. We are conducting a multidisciplinary investigation into the mechanisms of photoinduced electron transfer and proton transfer in GFP that incorporates spectroscopic tools developed in our laboratory.
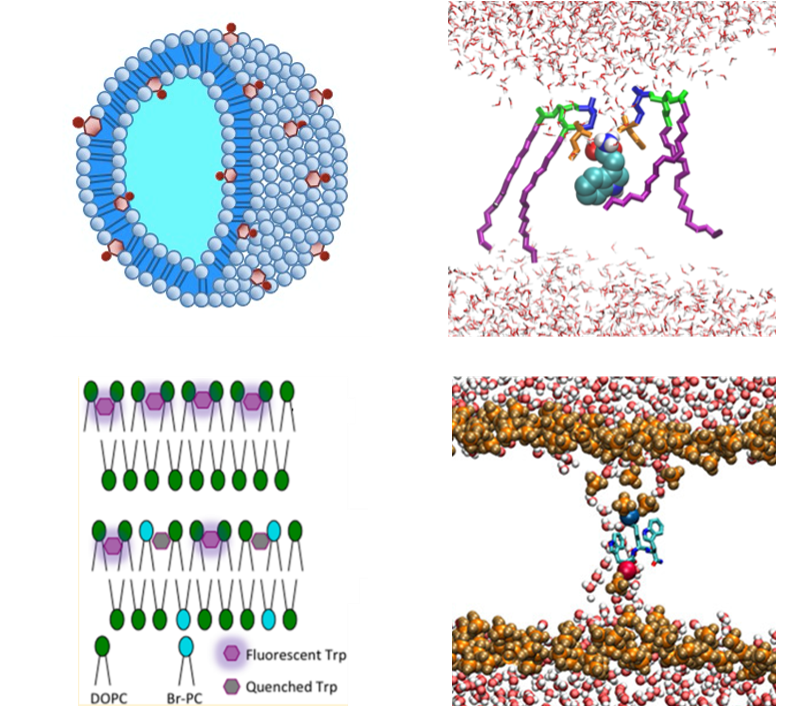
Lipid bilayer membranes control critical elements of biological function through structural and electrostatic mechanisms. Using a model peptide and characterization methods including FTIR, fluorescence, and molecular dynamics, we are investigating membranes at a molecular level to better understand the mechanism of self-assembly and function in the complicated and dynamic structures that they create.
